ESOMEPRAZOLE SODIUM
FOR INJECTION
PRODUCT DETAILS
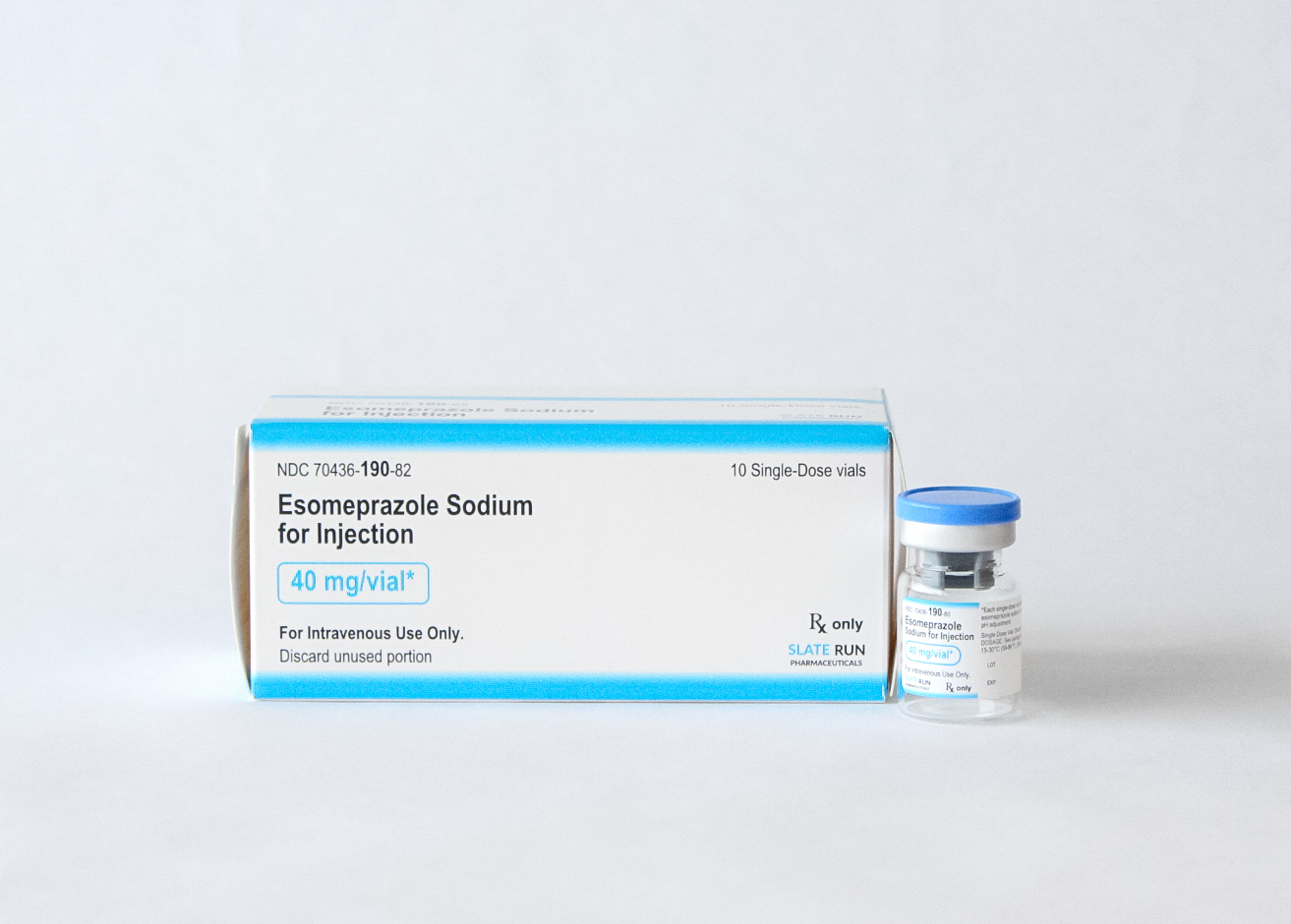
- Strength: Available in 40 mg* single-dose vials (*equivalent to 42.5 mg esomeprazole sodium)
- Fill Volume: Lyophilized powder in a 5 mL vial
- Therapeutic Class: Proton Pump Inhibitor
- Closure does not contain any natural rubber or latex
- Preservative Free
SALEABLE UNIT
40 mg per vial
5 mL single-dose vial
Box of 10 vials
NDC Number:
40 mg per vial: 70436-190-82
Available through your wholesaler:
NDC: 70436-190-82
Cardinal Health: 5783154
McKesson: 2614014
Amerisource: 10267389
| Strength | NDC | Saleable Unit |
|---|---|---|
| 40 mg/vial | 70436-190-82 | 5 mL vial Lyophilized Powder Box of 10 vials |
Esomeprazole sodium for injection is supplied in a single-dose vial as a white to off-white lyophilized powder for reconstitution containing 40 mg of esomeprazole.
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.] Protect from light. Store in carton until time of use.
Following reconstitution and administration, discard any unused portion of esomeprazole solution [see Dosage and Administration (2.4, 2.5)] .