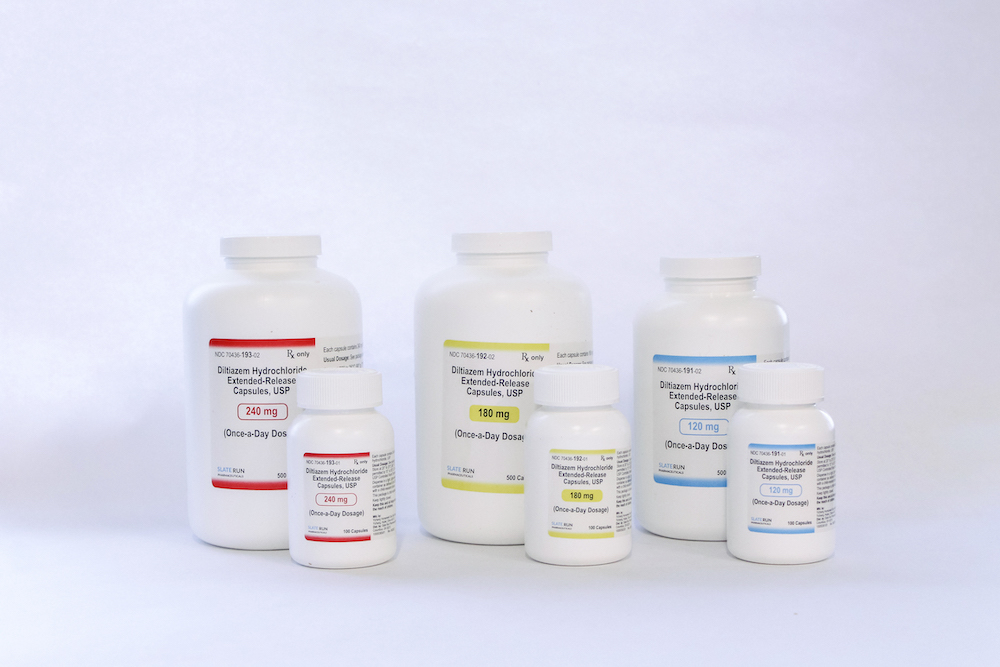
DILTIAZEM HYDROCHLORIDE
EXTENDED-RELEASE CAPSULES, USP
PRODUCT DETAILS
- Strength: Available in 120 mg,
180 mg, and 240 mg Capsules - Therapeutic Class: Cardiovascular Agent
NDC Numbers:
120 mg | Bottle of 100: 70436-191-01
180 mg | Bottle of 100: 70436-192-01
240 mg | Bottle of 100: 70436-193-01
Available through your wholesaler:
NDC: 70436-191-01
Cardinal Health: 5806823
McKesson: 2642023
Amerisource: 10272228
NDC: 70436-192-01
Cardinal Health: 5806849
McKesson: 2642049
Amerisource: 10272227
NDC: 70436-193-01
Cardinal Health: 5806864
McKesson: 2642064
Amerisource: 10272233
| Strength | NDC | Package Size |
|---|---|---|
| 120 mg | 70436-191-01 | Bottle of 100 |
| 180 mg | 70436-192-01 | Bottle of 100 |
| 240 mg | 70436-193-01 | Bottle of 100 |
Diltiazem Hydrochloride Extended-Release Capsules, USP are supplied as follows:
120 mg, opaque yellow cap and white body, imprinted with “A” and “148” on the cap in black ink.
180 mg, opaque light orange cap and white body, imprinted with “A” and “149” on the cap in black ink.
240 mg, opaque chocolate brown cap and white body, imprinted with “A” on the cap and “150” on the body in black ink.
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature].