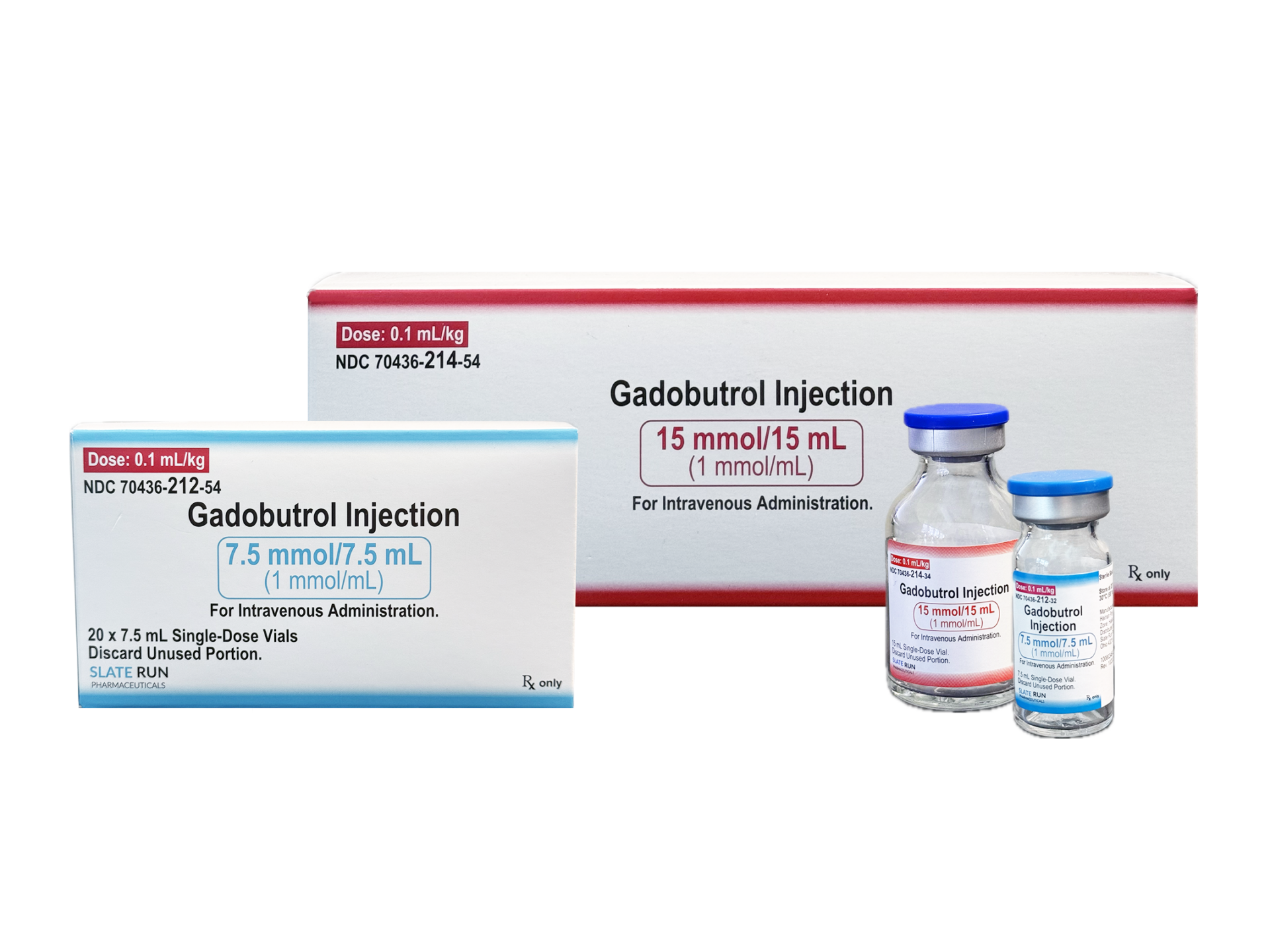
GADOBUTROL
INJECTION
PRODUCT DETAILS
- RLD: GADAVIST®
- Concentration:
1 mmol / 1 mL - Fill Volume: Sterile Solution in a
7.5 mL, 10 mL, 15 mL, 30 mL or 65 mL vial - Therapeutic Class: Contrast Agent
- Closure does not contain any natural rubber or latex.
- Preservative Free
NDC Numbers:
7.5 mL | Box of 20 vials: 70436-212-54
10 mL | Box of 10 vials: 70436-213-82
15 mL | Box of 20 vials: 70436-214-54
30 mL | Box of 5 vials: 70436-216-51
65 mL | Box of 1 vial: 70436-217-50
Available through your wholesaler:
NDC: 70436-212-54
FFF: GAD021254
Cardinal Health: 5908504
McKesson: 2919892
Amerisource: 10287497
NDC: 70436-213-82
FFF: GAD021382
Cardinal Health: 6055040
McKesson: 3061421
Amerisource: 10304250
NDC: 70436-214-54
FFF: GAD021454
Cardinal Health: 5908520
McKesson: 2919926
Amerisource: 50005963
NDC: 70436-216-51
FFF: GAD021651
Cardinal Health: 6055057
McKesson: 3061439
Amerisource: 10304112
NDC: 70436-217-50
FFF: GAD021750
Cardinal Health: 6055065
McKesson: 3061413
Amerisource: 10304049
| Strength | NDC | Saleable Unit |
|---|---|---|
| 7.5 mmol / 7.5 mL | 70436-212-54 | Box of 20 vials Sterile Solution (7.5 mL) |
| 10 mmol / 10 mL | 70436-213-82 | Box of 10 vials Sterile Solution (10 mL) |
| 15 mmol / 15 mL | 70436-214-54 | Box of 20 vials Sterile Solution (15 mL) |
| 30 mmol / 30 mL | 70436-216-51 | Box of 5 vials Sterile Solution (30 mL) |
| 65 mmol / 65 mL | 70436-217-50 | Box of 1 vials Sterile Solution (65 mL) |
Gadobutrol injection is a sterile, clear and colorless to pale yellow solution containing 604.72 mg gadobutrol per mL (equivalent to 1 mmol gadobutrol) per mL.
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature].
IMPORTANT SAFETY INFORMATION FOR:
GADOBUTROL INJECTION
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS
Risk Associated with Intrathecal Use
Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Gadobutrol injection is not approved for intrathecal use.
Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of gadobutrol injection in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs.
The risk for NSF appears highest among patients with:
- Chronic, severe kidney disease (GFR <30 mL/min/1.73m 2), or
- Acute kidney injury.
Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age >60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing.
For patients at highest risk for NSF, do not exceed the recommended gadobutrol injection dose and allow a sufficient period of time for elimination of the drug from the body prior to any re-administration.
Gadobutrol Injection is contraindicated in patients with history of severe hypersensitivity reactions to gadobutrol injection.
Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have uncommonly occurred following gadobutrol injection administration. Before gadobutrol injection administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and/or allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to gadobutrol injection. Administer gadobutrol injection only in situations where trained personnel and therapies are promptly available for the treatment of hypersensitivity reactions, including personnel trained in resuscitation.
Gadolinium is retained for months or years in several organs. While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies particularly closely spaced studies, when possible.
In patients with chronic renal impairment, acute kidney injury sometimes requiring dialysis has been observed with the use of some GBCAs. Do not exceed the recommended dose; the risk of acute kidney injury may increase with higher than recommended doses.
Most common adverse reactions (incidence ≥0.5%) are headache, nausea, and dizziness.
Please see the package insert for Gadobutrol Injection for full prescribing information.